Best Medical Device Quality Management Software Shortlist
Here’s my shortlist of the best medical device quality management software:
Managing quality in medical devices is no small feat. You're juggling compliance, document control, and product quality—all while ensuring you meet strict regulatory standards. I understand these challenges well.
In my experience, medical device quality management software can be a game-changer for your team. It helps you stay organized, maintain compliance, and focus on what truly matters: patient safety and product reliability.
In this article, I'll walk you through my top picks for medical device quality management software. I've independently tested and reviewed these tools to give you an unbiased look at what each offers. Let's find the right solution for your needs.
Why Trust Our Software Reviews
Best Medical Device Quality Management Software Summary
This comparison chart summarizes pricing details for my top medical device quality management software selections to help you find the best one for your budget and business needs.
| Tool | Best For | Trial Info | Price | ||
|---|---|---|---|---|---|
| 1 | Best for compliance tracking | Free demo available | Pricing upon request | Website | |
| 2 | Best for the pharmaceutical industry | Free demo available | Pricing upon request | Website | |
| 3 | Best for large enterprises | Free demo available | Pricing upon request | Website | |
| 4 | Best for flexible workflows | Free demo available | Pricing upon request | Website | |
| 5 | Best for life sciences companies | Free demo available | Pricing upon request | Website | |
| 6 | Best for cloud-based document control | Free demo available | Pricing upon request | Website | |
| 7 | Best for small to mid-sized teams | Free demo available | Pricing upon request | Website | |
| 8 | Best for medical device startups | Free demo available | Pricing upon request | Website | |
| 9 | Best for rapid deployment | Free demo available | Pricing upon request | Website | |
| 10 | Best for product-centric teams | Free demo available | Pricing upon request | Website |
Best Medical Device Quality Management Software Reviews
Below are my detailed summaries of the best medical device quality management software that made it onto my shortlist. My reviews offer a detailed look at the key features, pros and cons, integrations, and ideal use cases of each tool to help you find the best one for you.
ComplianceQuest is a cloud-based quality management system for industries focused on compliance and safety. It's used by quality managers and compliance officers to manage audits, track incidents, and ensure regulatory compliance. This tool helps your team stay on top of quality processes and requirements.
Why I picked ComplianceQuest: The software excels in compliance tracking, offering audit management and incident tracking capabilities. It provides a dashboard that allows you to monitor compliance status in real-time. Customizable workflows help your team adapt the tool to specific regulatory needs. Its analytics features provide insights into compliance trends and areas for improvement.
Standout features and integrations:
Features include audit management tools that simplify the auditing process, incident tracking to log and manage safety events, and a document control system to manage compliance documents. These features help your team maintain a high level of compliance and safety.
Integrations include Salesforce, SAP, Oracle, NetSuite, and more.
Pros and Cons
Pros:
- Detailed analytics and reporting
- Real-time compliance monitoring
- Customizable workflows available
Cons:
- High cost compared to competitors
- Complex initial configuration
Veeva Vault is a cloud-based content management and collaboration platform tailored for the pharmaceutical industry. It's used by regulatory teams to manage documents, ensure compliance, and simplify regulatory submissions. This tool helps your team efficiently handle complex document workflows and maintain compliance with industry standards.
Why I picked Veeva Vault: It caters specifically to the life sciences sector with features that support regulatory submissions. The software offers document management with automated version control and compliance tracking. You can manage submissions through its structured workflows, which are important for pharmaceutical companies. Its auditing capabilities ensure your team meets regulatory requirements.
Standout features and integrations:
Features include structured workflows for managing document approvals, compliance tracking to monitor regulatory standards, and audit trail capabilities to track document changes. These features help your team handle complex regulatory processes.
Integrations include Salesforce, SAP, Microsoft Office 365, and more.
Pros and Cons
Pros:
- Strong regulatory submission support
- Effective compliance tracking
- Tailored for pharmaceutical needs
Cons:
- High licensing and implementation costs
- Not ideal for non-regulated industries
MasterControl is an enterprise-level quality management software designed for regulated industries. It's used by large companies to manage quality processes, document control, and reduce operational risk. This tool helps your team maintain regulatory standards and simplify complex workflows.
Why I picked MasterControl: It offers extensive capabilities suited for large enterprises needing quality management. The software provides document control with automated workflows to handle complex processes. You can track compliance with its advanced analytics and notifications. Its scalable platform supports large teams with diverse patient data needs.
Standout features and integrations:
Features include analytics to monitor quality metrics, audit management for compliance, and training management to ensure your team is up to date. These features support your team's ability to manage quality.
Integrations are not currently listed by MasterControl.
Pros and Cons
Pros:
- Strong audit management
- Advanced document control
- Extensive analytics and reporting
Cons:
- Occasional system lag during updates
- Interface can feel dated and clunky
ETQ Reliance is a quality management software designed to manage compliance, document control, and risk management. This tool helps your team adapt workflows to specific business needs, providing flexibility in quality management.
Why I picked ETQ Reliance: It offers flexible workflows that allow you to tailor processes to fit your team's requirements. The software provides customizable dashboards and reporting tools that give you control over data presentation. You can automate quality tasks to reduce manual work and improve efficiency. Its risk management capabilities help your team identify and mitigate potential issues.
Standout features and integrations:
Features include tools to assess and mitigate risks, audit management to track compliance, and document control to keep all records organized. These features support your team in maintaining high-quality standards and compliance.
Integrations are not currently listed by ETQ Reliance.
Pros and Cons
Pros:
- Customizable dashboards and reports
- Automation reduces manual tasks
- Strong risk management capabilities
Cons:
- Slow interface performance
- High licensing and setup costs
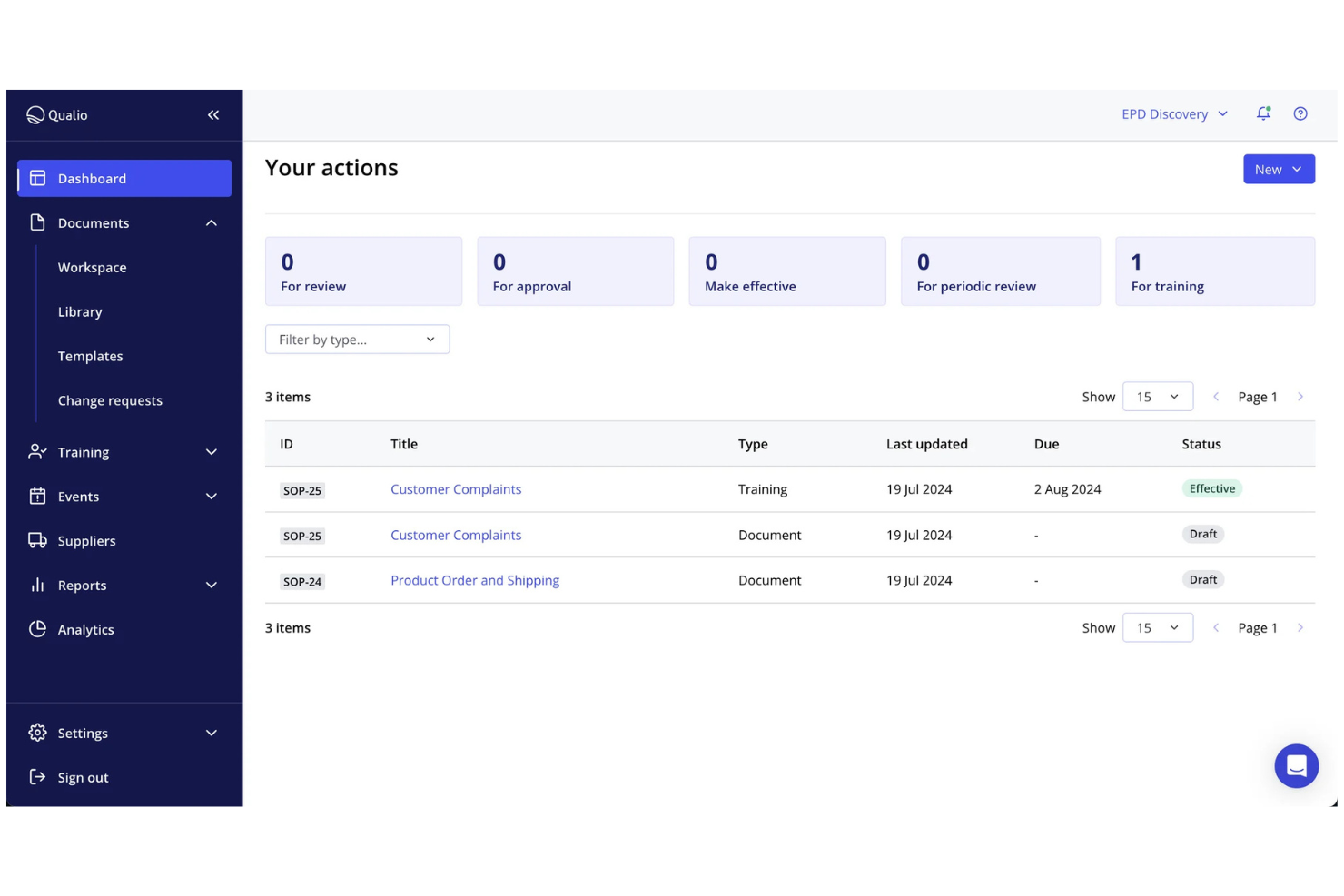
Qualio is a cloud-based quality management software designed for the life sciences industry, including medical device companies. It's used by quality assurance teams to manage compliance, document control, and training processes. This tool helps your team maintain quality, reduce enterprise risk, and maintain compliance standards.
Why I picked Qualio: It focuses on the life sciences sector and provides features tailored to industry needs. The software offers document management with automated version control, reducing manual errors. You can manage training programs to ensure your team meets compliance requirements. Its intuitive interface makes it accessible for teams new to digital quality systems.
Standout features and integrations:
Features include audit trail capabilities to track document changes, training records to keep your team compliant, and CAPA management to address quality issues. These features support your team in maintaining high compliance standards.
Integrations include Salesforce, Google Workspace, Okta, Jira, and more.
Pros and Cons
Pros:
- Effective training management
- Intuitive user interface
- Automated version control
Cons:
- Expensive for small startups
- Occasional slow loading speeds
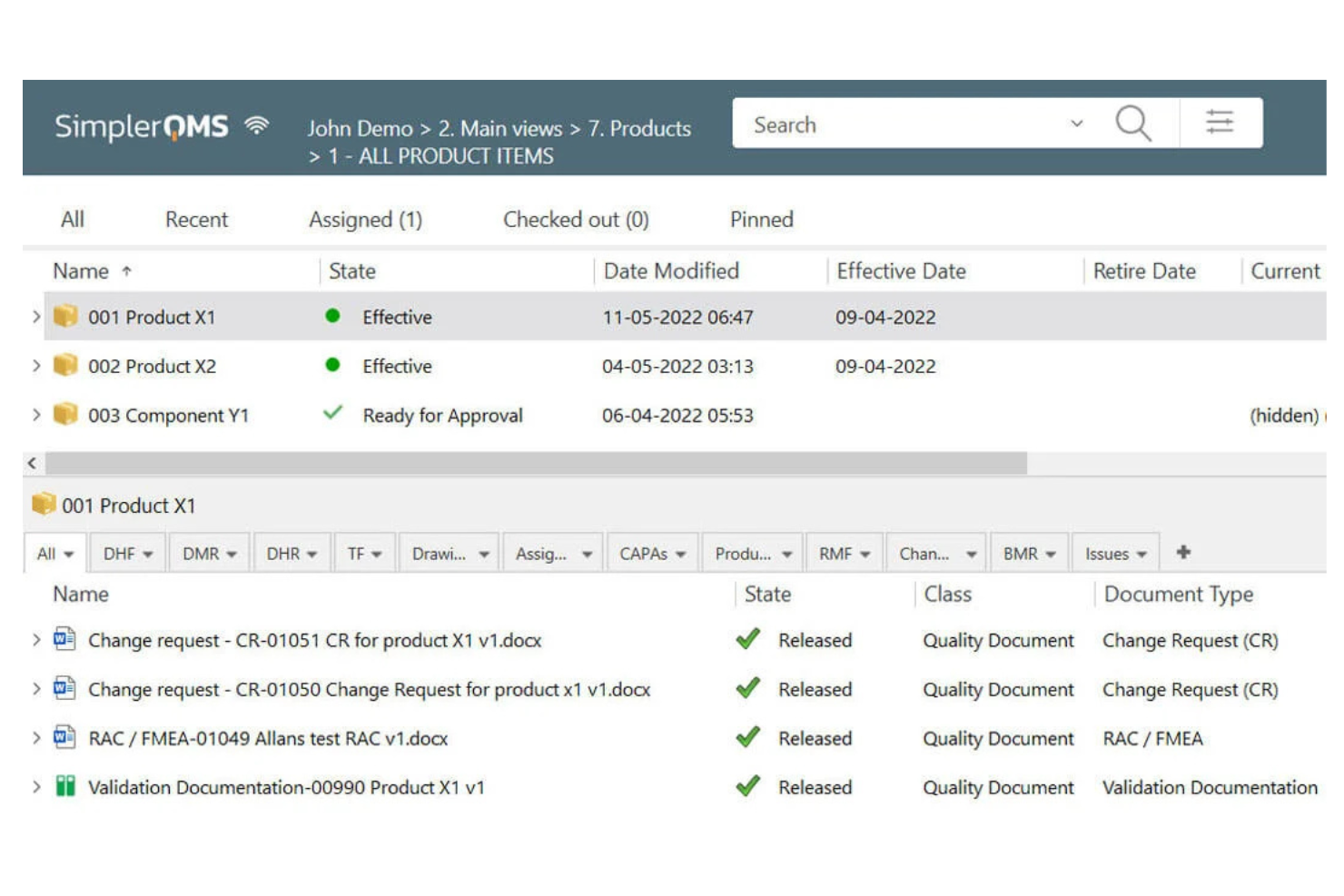
SimplerQMS is a cloud-based quality management system designed for life sciences and medical device companies. It's used by quality managers to organize and control documents, ensuring compliance with industry standards. This software helps your team manage quality processes efficiently in a cloud environment.
Why I picked SimplerQMS: It excels in cloud-based document control, offering an intuitive platform for managing compliance records. The software provides automated version control and electronic signatures, reducing manual paperwork. You can track changes and maintain an audit trail. These features make it ideal for teams needing efficient document management.
Standout features and integrations:
Features include training management to keep your team compliant, CAPA management to address non-conformances, and risk management to evaluate potential issues. These capabilities support your team's ability to meet regulatory standards.
Integrations include Microsoft Word, Microsoft SharePoint, Microsoft Teams, and more.
Pros and Cons
Pros:
- Effective audit trail tracking
- Supports electronic signatures
- Automated version control
Cons:
- Customization options are limited
- Mobile app lacks full features
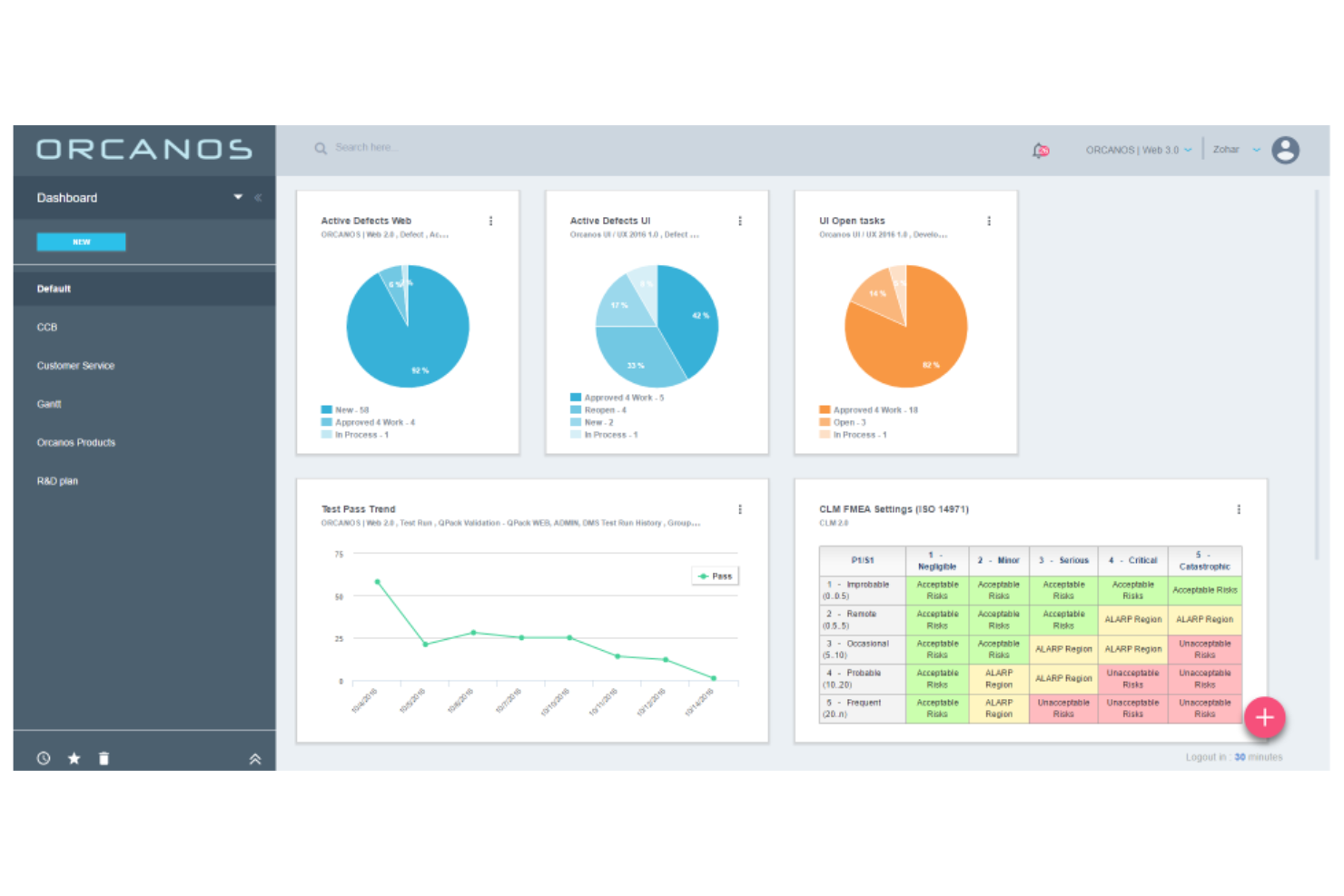
Orcanos is a quality management and compliance software tailored for medical device companies. It's mainly used by small to mid-sized teams to manage document control and ensure regulatory compliance. This tool helps your team optimize quality processes and maintain compliance.
Why I picked Orcanos: It offers a specialized solution for small to mid-sized teams, focusing on easy document control and compliance management. The software provides a user-friendly interface that simplifies document handling. You can track changes and maintain a clear audit trail for regulatory compliance. Its customizable workflows allow your team to adapt the software to specific needs.
Standout features and integrations:
Features include risk management tools to identify potential issues, CAPA management to address non-conformances, and training to keep your team compliant. These features support your team in maintaining high-quality standards.
Integrations are not currently listed by Orcanos.
Pros and Cons
Pros:
- User-friendly interface
- Customizable workflows
- Clear audit trail
Cons:
- Limited scalability for large teams
- Configuration can be complex
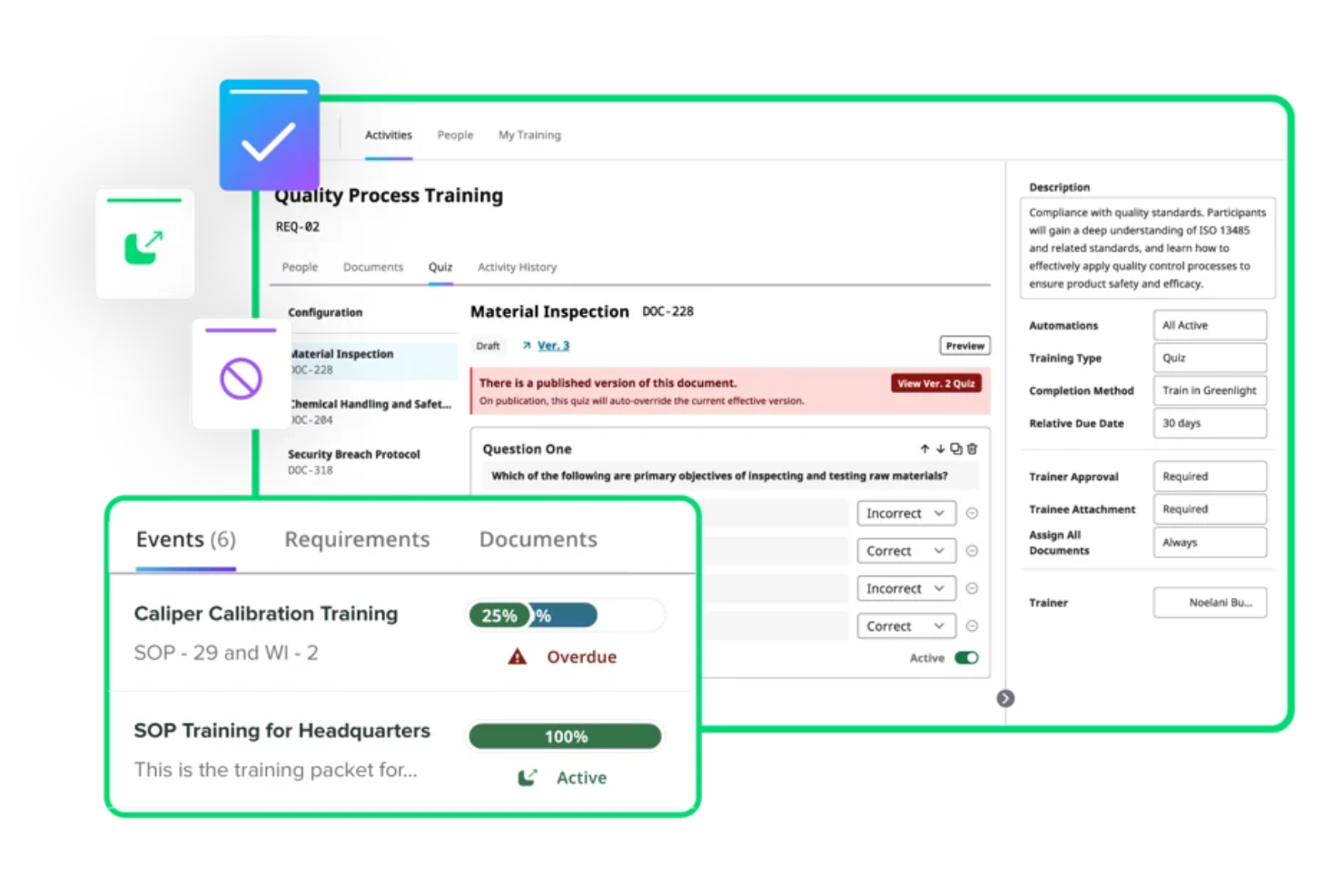
Greenlight Guru is an electronic Quality Management System (eQMS) tailored for medical device companies. It's primarily used by startups and small teams to manage compliance and quality processes efficiently. This tool helps you navigate product development while meeting regulatory requirements.
Why I picked Greenlight Guru: It offers a specialized platform for startups, focusing on minimal paperwork with its ultralight eQMS. The quality management software ensures compliance, which is important for new businesses in the medical field. Clinical data management features help collect compliant data for trials. These capabilities make it ideal for growing medical device companies.
Standout features and integrations:
Features include document and training management to keep your team updated, design and development tools for managing traceability, and quality processes for logging and resolving events. These features ensure your team stays compliant and efficient.
Integrations include Jira.
Pros and Cons
Pros:
- Supports clinical data management
- Strong compliance support
- Tailored for medical device startups
Cons:
- Limited scalability for large enterprises
- Limited integration options
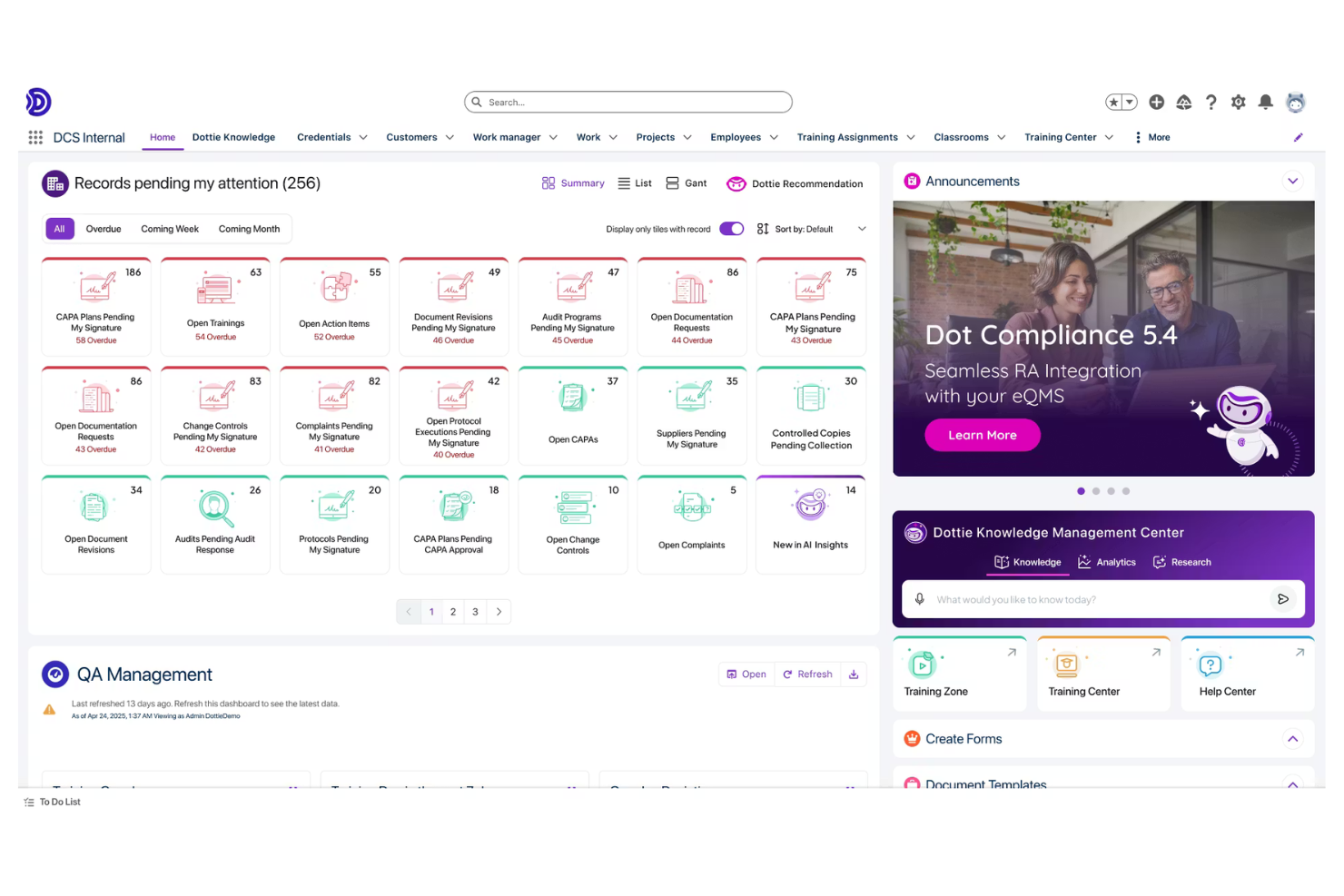
Dot Compliance is a quality management software designed for life sciences and medical device companies. It's used by quality assurance teams to manage compliance, document control, and quality processes. This tool helps your team quickly implement regulatory oversight systems and maintain compliance with industry standards.
Why I picked Dot Compliance: It offers rapid deployment capabilities, making it ideal for teams needing quick implementation. The software provides pre-configured solutions that reduce setup time and complexity. You can manage quality processes with its intuitive interface. Its built-in incident management features ensure your team meets regulatory requirements without extensive setup.
Standout features and integrations:
Features include pre-configured quality processes to minimize setup time, document control to manage HIPAA documents, and audit management to track health systems activities. These features support your team in maintaining high-quality standards with minimal delay.
Integrations include Salesforce, Microsoft Office 365, Google Workspace, and more.
Pros and Cons
Pros:
- Intuitive user interface
- Built-in compliance features
- Pre-configured solutions available
Cons:
- Limited customization options
- Requires a Salesforce license
Arena QMS is a cloud-based quality management system designed for product-centric teams, particularly in the medical device industry. It's used by quality and engineering teams to manage product development, quality process initiatives, and compliance. This tool helps your team integrate quality management into product design and development.
Why I picked Arena QMS: It offers a unified platform that connects quality management with product development. The software provides features such as design control and document management to support product-centric teams. You can optimize your product lifecycle with its collaborative platform, and the compliance management tools help you meet regulatory standards throughout product development.
Standout features and integrations:
Features include change management to track design revisions, training administration to ensure team compliance, and quality process automation to reduce manual tasks. These features support your team's ability to maintain high-quality standards in product development.
Integrations include Salesforce, Oracle, SAP, Microsoft Dynamics, Jira, SolidWorks, and more.
Pros and Cons
Pros:
- Supports quality and compliance management
- Effective design control features
- Strong product lifecycle integration
Cons:
- Integration with legacy tools is hard
- Not ideal for non-product teams
Other Medical Device Quality Management Software
Here are some additional medical device quality management software options that didn’t make it onto my shortlist, but are still worth checking out:
- Formwork
For low-cost solutions
- Matrix One
For traceability in development
- QT9 Software
For automated compliance oversight
Medical Device Quality Management Software Selection Criteria
When selecting the best medical device quality management software to include in this list, I considered common buyer needs and pain points, such as ensuring regulatory compliance and managing document control efficiently. I also used the following framework to keep my evaluation structured and fair:
Core Functionality (25% of total score)
To be considered for inclusion in this list, each solution had to fulfill these common use cases:
- Document control and management
- Compliance with regulatory standards
- Audit management
- Risk management
- CAPA (Corrective and Preventive Action) management
Additional Standout Features (25% of total score)
To help further narrow down the competition, I also looked for unique features, such as:
- Customizable workflows
- Integration with other business systems
- Advanced analytics and reporting
- Cloud-based accessibility
- Electronic signatures
Usability (10% of total score)
To get a sense of the usability of each system, I considered the following:
- Intuitive user interface
- Ease of navigation
- Minimal training required
- Aesthetic design
- Responsive design for mobile devices
Onboarding (10% of total score)
To evaluate the onboarding experience for each platform, I considered the following:
- Availability of training videos
- Interactive product tours
- Step-by-step onboarding guides
- Access to webinars and tutorials
- Templates for quick setup
Customer Support (10% of total score)
To assess each software provider’s customer support services, I considered the following:
- 24/7 support availability
- Multiple support channels (chat, email, phone)
- Response time to inquiries
- Availability of a knowledge base
- Personalized support options
Value For Money (10% of total score)
To evaluate the value for money of each platform, I considered the following:
- Competitive pricing
- Feature set versus cost
- Flexible pricing plans
- Discounts for long-term contracts
- Cost of add-ons and integrations
Customer Reviews (10% of total score)
To get a sense of overall customer satisfaction, I considered the following when reading customer reviews:
- Overall satisfaction ratings
- Feedback on software reliability
- Comments on ease of use
- Reports on customer support experiences
- Testimonials on value for money
How To Choose Medical Device Quality Management Software
It’s easy to get bogged down in long feature lists and complex pricing structures. To help you stay focused as you work through your unique software selection process, here’s a checklist of factors to keep in mind:
| Factor | What To Consider |
|---|---|
| Scalability | Can the software grow with your company? Consider future expansion and whether the tool can handle increased data and users without compromising performance. |
| Integrations | Does it work with your existing systems? Check compatibility with current tools such as ERP, CRM, and document management systems to avoid workflow disruptions. |
| Customizability | Can you tailor it to your needs? Look for flexibility in workflow configurations to match your specific processes and unique business needs. |
| Ease of Use | Is it user-friendly for your team? Evaluate the learning curve and whether your team can navigate the interface without extensive training. |
| Implementation and Onboarding | How long before it's fully operational? Assess the time and resources needed for setup and employee training to minimize disruptions in daily operations. |
| Cost | Is it within your budget? Analyze total cost of ownership, including subscription fees, support, training, and potential add-ons, to avoid hidden expenses. |
| Security Safeguards | Does it protect your data? Ensure compliance with industry standards such as ISO 27001 and check for features such as data encryption and access controls. |
| Compliance Requirements | Does it meet regulatory standards? Verify alignment with necessary regulations such as FDA, ISO, and EU MDR to avoid compliance issues. |
What Is Medical Device Quality Management Software?
Medical device quality management software is a tool used to ensure compliance with regulatory standards and manage quality processes in medical device manufacturing. It's generally used by quality assurance professionals, regulatory affairs specialists, and compliance teams to maintain product quality and safety.
Document control, risk management, and audit trail capabilities help with regulatory compliance and efficient quality management. These tools provide significant value by reducing errors, ensuring compliance, and supporting the overall quality of medical devices.
Features
When selecting medical device quality management software, keep an eye out for the following key features:
- Document control: Manages and organizes all documents related to quality processes, ensuring easy access and compliance with healthcare industry standards.
- Risk management: Identifies, assesses, and mitigates potential risks in product development and manufacturing, helping maintain safety and compliance.
- Audit management: Tracks and manages audits to ensure compliance with industry regulations and standards, providing a clear audit trail.
- CAPA management: Facilitates corrective and preventive actions to address non-conformances, improve product quality, and avoid adverse patient outcomes.
- Compliance tracking: Monitors and ensures adherence to industry regulations, reducing the risk of non-compliance and associated penalties.
- Training management: Manages employee training modules to ensure staff are up to date with compliance and quality standards.
- Quality process automation: Handles repetitive quality tasks, improving efficiency and reducing manual errors.
- Version control: Keeps track of document revisions, provides cybersecurity, and ensures that the current version is accessible, supporting consistency and accuracy.
- Customizable workflows: Allow users to tailor workflows to specific healthcare operations needs, providing flexibility and efficiency.
- Analytics and reporting: Provides risk data insights into quality metrics and performance, helping teams make informed decisions and improvements.
Benefits
Implementing medical device quality management software provides several benefits for your team and your business. Here are a few you can look forward to:
- Improved compliance: Ensures adherence to industry regulations through compliance tracking and audit management, reducing the risk of penalties.
- Improve efficiency: Automates quality processes such as document control and CAPA management, saving time and reducing adverse events.
- Better risk management: Identifies and provides risk management solutions early in the product lifecycle, helping maintain product safety and quality.
- Increased transparency: Provides clear audit trails and version control, making it easier to track changes and maintain accountability to support patient care.
- Efficient training: Manages training programs to keep your team updated with the latest compliance and quality standards.
- Data-driven decisions: Offers analytics and reporting tools that provide insights into quality metrics, supporting informed decision-making.
Costs and Pricing
Selecting medical device quality management software requires an understanding of the various pricing models and plans available. Costs vary based on features, team size, add-ons, and more. The table below summarizes common plans, their average prices, and typical features included in medical device quality management software solutions:
Plan Comparison Table for Medical Device Quality Management Software
| Plan Type | Average Price | Common Features |
|---|---|---|
| Free Plan | $0 | Basic document management, limited support, and simple compliance tracking. |
| Personal Plan | $10000-$50000/year | Document control, basic risk management, and audit trail capabilities. |
| Business Plan | $20000-$200000/year | Advanced compliance tracking, CAPA management, and customizable workflows. |
| Enterprise Plan | $100000+/year | Full feature set including analytics, extensive integrations, and premium support. |
Medical Device Quality Management Software FAQs
Here are some answers to common questions about medical device quality management software:
What industries use medical device quality management software?
Medical device quality management software is primarily used in the healthcare and life sciences industries, including medical device manufacturing, pharmaceuticals, and biotech companies. It helps teams in these sectors manage healthcare compliance with regulations such as FDA and ISO standards, ensuring product quality and safety.
How do I know if my business needs medical device quality management software?
If your business operates in a regulated industry with strict policy management requirements, you likely need medical device quality management software. It’s essential for managing documentation, tracking healthcare compliance, and ensuring quality standards. Consider factors such as regulatory challenges, team size, and product complexity to determine your need.
Can medical device quality management software integrate with existing systems?
Yes, most medical device quality management software can integrate with existing systems such as ERP, CRM, and document management tools. When evaluating software, check for compatibility with your current systems to ensure smooth workflows. Integrations can improve efficiency, eliminate data silos, and reduce manual entry.
Is training required to use medical device quality management software?
Yes, training is usually required to effectively use medical device quality management software. Most vendors offer resources such as training videos, webinars, case studies, and product tours to help your team get up to speed. Investing time in training can maximize the software’s benefits and improve team efficiency.
What's Next:
If you're in the process of researching medical device quality management software, connect with a SoftwareSelect advisor for free recommendations.
You fill out a form and have a quick chat where they get into the specifics of your needs. Then you'll get a shortlist of software to review. They'll even support you through the entire buying process, including price negotiations.